A person wearing the VidaSense band, illustrating how the device fits closely to the body without gels, wires, or external attachments.
A wearable approach shaped around care
VidaSense is developing a wearable monitoring solution based on non-invasive fetal electrocardiogram (NIfECG) technology. During labor, the system is designed to continuously capture key maternal-fetal signals, including fetal heart rate, maternal heart rate, and uterine activity, while reducing sensitivity to sensor positioning and the need for repeated bedside adjustment. Unlike conventional ultrasound-based systems, the VidaSense approach uses dry electrodes and does not require gel for signal acquisition, helping make monitoring more practical and less restrictive.
“Too often, fetal monitoring still asks mothers to adapt to the technology,” says VidaSense Founder Naeimeh Behbood. “Our goal with VidaSense is to turn that around and create a solution that fits the realities of pregnancy and labor.”
That patient-centered logic is central to the project. In women’s health, comfort is not separate from performance. It is part of whether a solution can be used consistently, accepted in practice, and integrated into care.

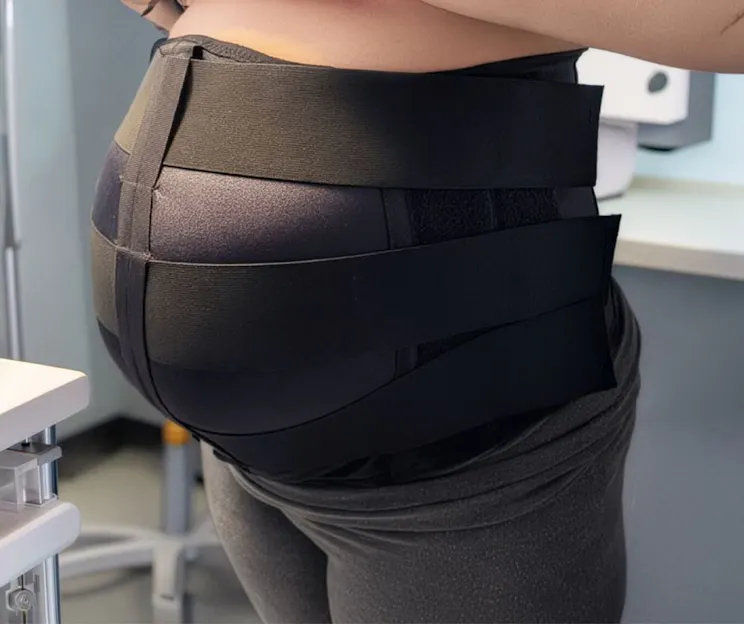
Another view of the VidaSense band, highlighting its streamlined wearable design with no gels, additional wiring, or external components.
Why labor is the decisive test
What makes this phase especially significant is the setting. The clinical testing takes place in labor wards of the Women's Hospital, Inselspital, Bern University Hospital. The study is designed for fetal–maternal monitoring in two distinct settings: during antepartum in the third trimester and during the first stage of labor. It will evaluate signal quality, measurement accuracy, and maternal comfort.
The project also draws on clinical know-how in obstetrics and maternity care. The collaboration includes specialists such as Prof. Dr. med. Daniel Surbek, Head of Obstetrics and Feto‑Maternal Medicine, Acting Co-Chairman of the Women’s Hospital, Inselspital, Bern University Hospital, and PD Dr. med. Anda Radan, Head of the Obstetrics Outpatient Department, Women's Hospital, Inselspital, Bern University Hospital, as well as midwifery expertise. That multidisciplinary combination matters because translating a wearable monitoring concept into practice depends not only on engineering, but also on how well the system fits labor-room realities and clinical decision-making.
If the system performs well in this setting, it strengthens the case for clinical relevance and future adoption.

Close-up of the VidaSense technology integrated into the wearable band, showing the compact dry electrode sensing technology embedded within the device.
From research to spin-off
VidaSense did not emerge in isolation. It builds on a longer CSEM innovation pathway in dry electrodes, wearable monitoring, and remote prenatal care. These earlier research projects helped establish the technology base from which the spin-off emerged.
The spin-off also gained momentum through ACCELERATE, CSEM’s start-up program, before entering its current clinical phase through the Bern Medtech Collaboration Call. That continuity is what gives the story its depth.
“What makes this project particularly strong is the way it connects deep-tech research, spin-off creation, and clinical study,” says Jens Krauss, VP of Medtech at CSEM. “That is how innovation moves closer to becoming a real medical product.”
The project also highlights the strength of the Bern medtech ecosystem, bringing together CSEM, clinical expertise, academic partners, and start-up-driven translation.

Additional Information
Discover more about VidaSense through their website.
This innovation project is supported by Innosuisse Project Number 122.849 IP-LS.



